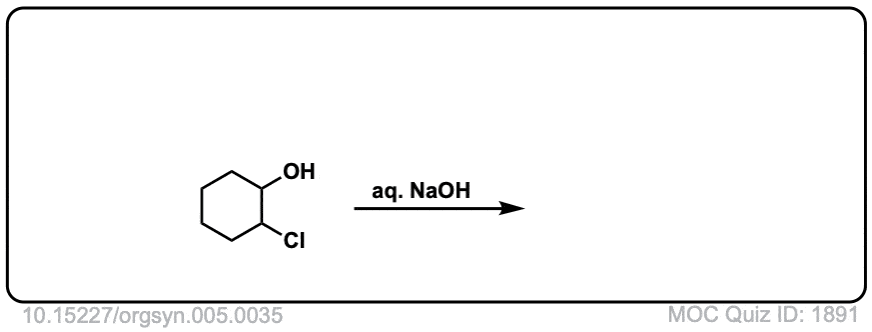
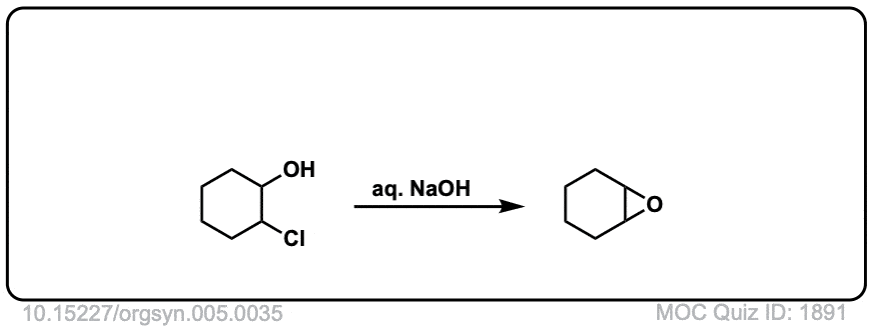
Formation of epoxides from bromohydrins
Description: Halohydrins treated with strong base (such as hydroxide ion) can form epoxides through an intramolecular SN2 reaction.
The rest of this page is available to MOC Members only.
To get access to this page, plus over 1500 quizzes, the Reaction Encyclopedia, Org 1 / Org 2 summary sheets, and flashcards, sign up here for only 30 cents/ day!
Real-Life Example:
Org. Synth. 1925, 5, 35
DOI Link: 10.15227/orgsyn.005.0035
 Click to Flip
Click to Flip
Could you please include the mechanism for example 4 where there is no epoxide formation due to reagents being cis to eachother. Thanks.
I might be looking at the wrong page, but what about forming an epoxide from a reagent containing Cl (and no alkene) that gives a product with no stereochemistry displayed? Is this possible?
Hi Katherine – Thanks for your comment! Sorry for the late response! I can’t picture what you’re describing. Do you have a pic somewhere?
Maybe you were looking for formation of epoxides using m-chloroperoxybenzoic acid? Or something completely different?
If you don’t get back to me no worries but I try to answer comments!
James
Is it possible for a trans-1-chlorocyclopentan-2-ol to react with sodiumhydride and to form a epoxide and hydrogengas? Or does it not react in this way because of the steric hinderence and wil it form something else or will it not react at all?
If someone could help me out that will be awesome?
Greetings,
Miguel
Yes, sodium hydride is a base, and it would deprotonate the alcohol to give an alkoxide and then form an epoxide.
Maybe I misssed something but I don’t understand why in the first example we get alkane instead of epoxide ?
Could you explain what is the reason?
Greetings
Maybe I misssed something but I don’t understand why in the first example we get alkane instead of epoxide ?
Could you explain what is the reason?
Greetings
It’s a typo. Thanks for pointing it out! Fixed.